【Anode】Understanding Graphite Anodes in One Article

The rapid growth of the EV and energy storage industries is boosting demand for high-performance lithium batteries, driving the market for quality petroleum coke and synthetic graphite. The quality and particle size of calcined petroleum coke directly affect synthetic graphite performance, especially in anode production.
【Anode】Understanding Graphite Anodes in One Article
Lithium-ion batteries (LiBs) power electric vehicles (EVs), and the anode plays a crucial role in determining their performance. Graphite materials, with their excellent electrical conductivity, thermal stability, and high performance, are the primary anode materials for lithium batteries. However, graphite anodes still suffer from initial capacity loss and limited first-cycle efficiency. The most common solution is to coat graphite particles, i.e., to add a protective layer that prevents undesirable chemical reactions during battery cycling and improves overall performance.
Preface
Over the past three decades, lithium-ion batteries (LIBs) have undergone significant evolution, transitioning from powering small devices to enabling large-scale applications such as electric vehicles (EVs) and stationary energy storage systems. A key advancement in this process has been the adoption of graphite-based anodes, replacing soft carbon and hard carbon. Due to graphite’s low lithiation/delithiation potential and its impressive theoretical gravimetric capacity of 372 mAh/g, this transition has greatly improved the overall energy density of full cells [1].
As shown in Figure 1, improvements in graphite electrodes have a long history. Since 1975, it has been known that graphite can form a compound with lithium through a reversible process, namely LiC₆. However, in the 1970s, due to issues with liquid organic electrolytes, graphite could not be successfully used in batteries, leading to continuous decomposition. After 20 years of research and development on graphite anodes, Sony introduced the first lithium-ion battery in 1991, marking a major breakthrough in battery technology [2].
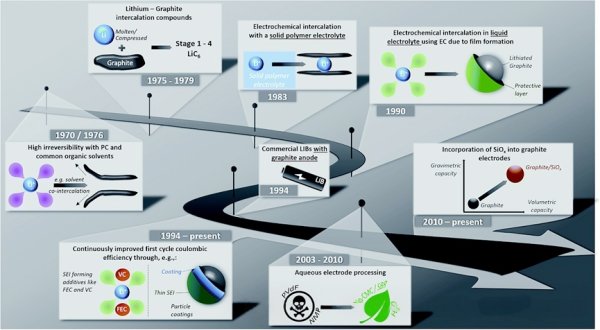
Figure 1. Major milestones in the development of graphite anodes for lithium-ion batteries [2].
Since 1994, most commercial lithium-ion batteries have used graphite as the anode active material due to its low cost, relatively high theoretical gravimetric capacity (372 mAh/g), and high coulombic efficiency. According to the 2015 anode material market share, approximately 98% of lithium battery anodes are made from carbon-based materials. The remaining 2% consists of silicon (Si) and lithium titanate (LTO). Major EV and battery manufacturers believe that graphite will continue to be a key component in lithium batteries, either as a sole anode or in combination with other elements such as silicon (Si) [1,2].
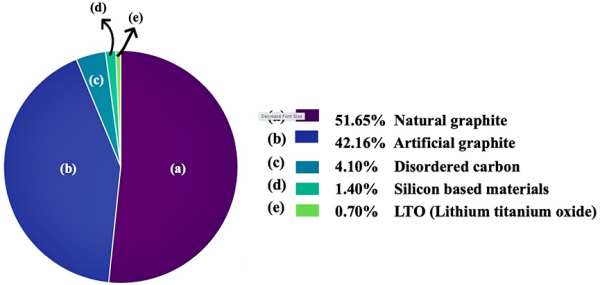
Figure 2. Market share of anode materials in 2015 [1].
What is Graphite?
Carbon is the sixth element in the periodic table, with a molar mass of 12.01 g/mol. In nature, carbon materials mainly exist in two structures: diamond and graphite. In graphite, sp²-hybridized graphene layers are bonded together by weak van der Waals forces. As shown in Figure 3(a), this bonding results in a hexagonal atomic arrangement, forming layered sheets with an interlayer spacing of 3.354 Å. The relatively weak van der Waals forces between adjacent graphene layers facilitate the intercalation of ions and molecules between these layers, forming a two-dimensional layered structure. These forces also allow expansion between graphene layers, enabling restacking and reversible lithium-ion intercalation. This intercalation reaction exhibits excellent reversibility with minimal volume change, ensuring long-term efficiency and reliability in lithium-ion batteries and highlighting the importance of graphite as an anode material [2].
Due to its layered arrangement, graphite particles typically exhibit a flat, flake-like morphology with two distinct surfaces: the basal plane with dimension La and the edge plane with height Lc, also known as the prismatic plane. This is illustrated schematically in the SEM (scanning electron microscope) images in Figures 3 and 4. As a result, the edge plane shows higher intercalation/deintercalation reactivity compared to the basal plane [3].
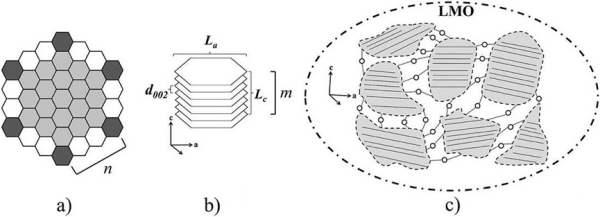
Figure 3. Schematic diagram of graphite crystal structure [3].
The edge planes in graphite are widely recognized as active sites that play a critical role in various reactions. However, these edge sites are a double-edged sword, as they can also trigger undesirable reactions that affect system efficiency. The electrochemical performance of graphite electrodes largely depends on the ratio of basal planes to edge planes. While edge planes facilitate intercalation, excessive edge planes can increase surface reactivity, including side reactions or long-term degradation, thereby compromising battery stability and cycle life. Notably, different types of graphite may exhibit variations in the ratio of basal to edge planes, influencing overall battery efficiency, charge/discharge characteristics, and suitability for specific applications. This underscores the importance of tailoring graphite structures to optimize electrochemical performance in various battery systems [1,2].

Figure 4. SEM image of graphite flakes [2].
Solid Electrolyte Interphase (SEI)
As mentioned earlier, irreversible capacity loss originates from electrolyte decomposition, leading to undesirable reactions and the formation of a solid electrolyte interphase (SEI) on the graphite surface. Common electrolytes used in lithium-ion batteries, such as propylene carbonate (PC) and ethylene carbonate (EC), react strongly with graphite to form the SEI layer. While the SEI is essential for stabilizing the electrode–electrolyte interface, it also reduces overall battery capacity due to irreversible lithium loss during its formation. Unfortunately, SEI formation is not limited to the first cycle; it continues to evolve during battery operation due to ongoing interactions with the electrolyte. During cycling, graphite undergoes slight volume expansion due to intercalation/deintercalation, leading to microcracks in the SEI. These microcracks expose new surfaces, promoting further SEI formation and resulting in cumulative capacity loss over time. Among various strategies, coating has emerged as the most effective method to reduce microcrack formation and address initial capacity loss in graphite anodes [1,2].
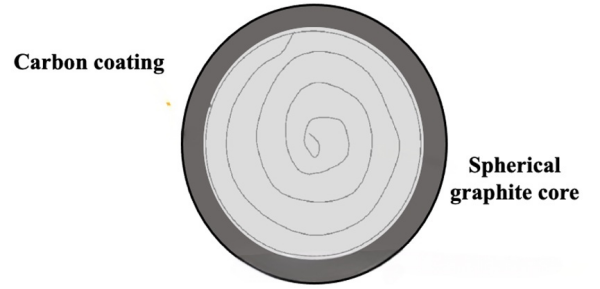
Figure 5. Coated spherical graphite [2].
Graphite Coating
The properties of the SEI largely depend on the electrolyte composition and the surface characteristics of graphite. Surface modification methods, such as coating, can promote the early formation of a stable SEI. This stable interface prevents undesirable reactions and is crucial for improving cycle performance and initial coulombic efficiency (ICE). In 1996, Peled et al. [4] first demonstrated the effectiveness of surface modification in enhancing performance by mildly oxidizing graphite particles at 550°C for 1 hour, resulting in a 10% increase in initial coulombic efficiency.
Various precursors, including metals, gases, and solid carbon sources, have been used to modify graphite surfaces in lithium batteries. To date, carbon has been the most widely studied and cost-effective coating material, applicable not only to graphite but also to cathode and anode materials. In this process, graphite is initially coated with a saturated carbon precursor such as glucose. To stabilize the coating, high-temperature treatment (e.g., 900°C) is applied to carbonize the precursor-coated material. By covering reactive sites on the graphite nanostructure, carbon coatings mitigate adverse interactions between the SEI and the electrolyte. However, compared to graphite, these carbon coatings have lower density and significantly reduced energy storage capacity, which can lead to lower gravimetric and volumetric energy density at the full-cell level.
Among carbon-based agents, coal tar pitch (CTP) is the standard industrial coating material, capable of forming a uniform amorphous carbon layer on graphite surfaces while improving capacity, rate performance, and initial coulombic efficiency. According to Han et al. [5], CTP-coated graphite exhibits an initial coulombic efficiency of 90.3% and superior rate capability. Despite its favorable electrochemical properties, CTP poses environmental and health concerns. It is derived from coal tar, a by-product of coal carbonization used in coke or gas production—a carbon-intensive process that releases polycyclic aromatic hydrocarbons (PAHs) into the environment. Additionally, the U.S. National Cancer Institute has reported that exposure to CTP increases the risk of skin cancer and is associated with other cancers, including lung, bladder, kidney, and gastrointestinal cancers [6].
Future Work
Researchers are currently focused on developing the most environmentally friendly advanced graphite anode coating materials for lithium-ion batteries. The goal is to use biomass-derived carbon coatings to produce high-performance anodes that can match or exceed the performance of CTP-coated graphite while minimizing health and environmental impacts. Although biomass coatings face challenges such as higher inorganic impurity content and variable performance, the drive toward sustainability continues to motivate improvements in battery materials. While CTP offers consistency, its non-renewable nature highlights the need to explore alternatives with lower environmental footprints [2].
Conclusion
The unique layered structure of graphite makes it highly suitable for lithium-ion intercalation. Since 1994, nearly all commercial lithium-ion batteries have used graphite as the anode active material. Despite its excellent conductivity and stability, graphite anodes suffer from significant first-cycle capacity loss due to electrolyte decomposition and uncontrolled SEI formation. Surface coating of graphite is one of the most effective solutions to this issue. Balancing cost and complexity while enhancing battery stability, efficiency, and capacity is key to advancing graphite anode technology. Among the materials tested, disordered carbon coatings have proven most effective in preventing undesirable reactions and reducing initial capacity loss. Coal tar pitch (CTP) remains the standard industrial coating but has significant environmental and health impacts. Current research focuses on developing greener alternatives, such as biomass-derived carbon coatings, to achieve high electrochemical performance with minimal adverse effects.
References
[1] Warner, J.T., 2019. Lithium-ion battery chemistries: a primer. Elsevier.
[2] Asenbauer, J., Eisenmann, T., Kuenzel, M., Kazzazi, A., Chen, Z. and Bresser, D., 2020. The success story of graphite as a lithium-ion anode material—fundamentals, remaining challenges, and recent developments including silicon (oxide) composites. Sustainable Energy & Fuels, 4(11), pp.5387–5416.
[3] Ouzilleau, P., Gheribi, A.E. and Chartrand, P., 2016. The graphitization temperature threshold analyzed through a second-order structural transformation. Carbon, 109, pp.896–908.
[4] Peled, E., Menachem, C., Bar-Tow, D. and Melman, A., 1996. Improved graphite anode for lithium-ion batteries: chemically bonded solid electrolyte interface and nanochannel formation. Journal of The Electrochemical Society, 143(1), p.L4.
[5] Han, Y.J., Kim, J., Yeo, J.S., An, J.C., Hong, I.P., Nakabayashi, K., Miyawaki, J., Jung, J.D. and Yoon, S.H., 2015. Coating of graphite anode with coal tar pitch as an effective precursor for enhancing rate performance in Li-ion batteries: effects of composition and softening points of coal tar pitch. Carbon, 94, pp.432–438.
[6] Jang, T.W., Kim, Y., Won, J.U., Lee, J.S. and Song, J., 2018. Standards for recognition of occupational cancers related to polycyclic aromatic hydrocarbons (PAHs) in Korea. Annals of Occupational and Environmental Medicine, 30, pp.1–6.
Feel free to contact us anytime for more information about the Anode Material market. Our team is dedicated to providing you with in-depth insights and customized assistance based on your needs. Whether you have questions about product specifications, market trends, or pricing, we are here to help.
No related results found








0 Replies